Posted on September 22, 2016 in Structural Biology Initiative
CUNY Advanced Science Research Center’s Amedee des Georges is co-first author on a paper shedding light on the structure and function of an intracellular receptor which controls the contraction of skeletal muscle, the findings of which are published today in Cell.
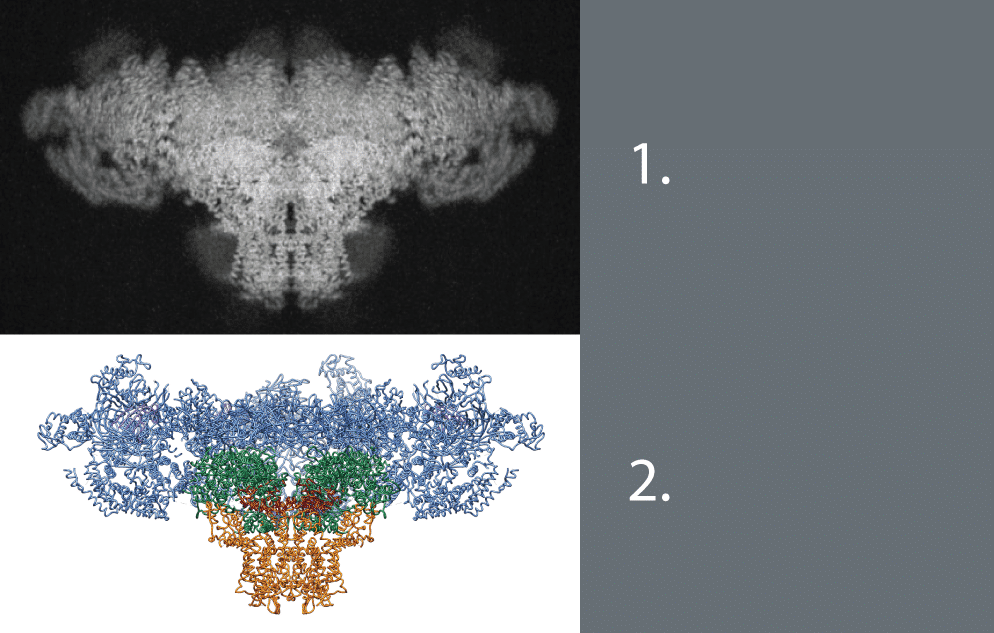
Des Georges’s role in the research began while a Howard Hughes postdoctoral associate at the Columbia University Medical Center (CUMC) and continued after being appointed as assistant professor with the CUNY ASRC’s Structural Biology Initiative. The paper—entitled “Structural basis for gating and activation of RyR1”—may pave the way to new techniques for treating heart and selected muscle disorders.
“These are very exciting findings and show the power of cryo-electron microscopy for looking at the function and dynamics of these molecules and for rational drug design,” des Georges said.
Utilizing high-resolution cryo-electron microscopy, the team—led by Howard Hughes Medical Institute (HHMI) Investigator Joachim Frank together with Andrew R. Marks, MD and Wayne A. Hendrickson, professors at CUMC—identified previously unobserved states of the Ryanodine receptors (RyRs), ion channels which control muscle contraction in skeletal muscle cells as well as heart muscle cells.
When the RyRs are activated, the receptors open and release calcium into the cell body, thus triggering muscle fiber contraction. The team’s previous work determined the 3D structure of an RyR in its closed state, but this new work provides a glimpse of the structure in its open state in addition to other conformations where the structure has changed shape, but not been opened.
This new information gives researchers a better understanding of the dynamics of the molecule and how alterations in that dynamics might lead to a variety of conditions, including age-related muscle weakness, muscular dystrophy and heart failure. Additionally, the findings of the paper will allow for further development of drugs which can restore RyR function.
This is des Georges’ second paper to appear in Cell, and the first since he joined the ASRC. Initiative Director Kevin Gardner said that this is a concrete sign of the exciting start of des Georges’ independent research program.
“This is a great example of an outstanding young structural biologist taking advantage of the latest technological advances to answer long-standing questions in biology,” Gardner said. “Given Amedee’s hard work and intuition, coupled with the excellent environment here at the ASRC and our partners nearby at CCNY and the New York Structural Biology Center, I anticipate that it’ll be the first of many such examples from him and SBI overall.”
To read the paper, please click here. For more information about the CUNY ASRC Structural Biology Initiative, please visit their website.
###
The City University of New York’s Advanced Science Research Center (ASRC) is a University-wide venture that elevates CUNY’s legacy of scientific research and education through initiatives in five distinctive, but increasingly interconnected disciplines: Nanoscience, Photonics, Structural Biology, Neuroscience and Environmental Sciences. The center is designed to promote a unique, interdisciplinary research culture with researchers from each of the initiatives working side by side in the ASRC’s core facilities, sharing equipment that is among the most advanced available.
###
